Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Supports Healthy Joint Function in Horses
Product Description
NexHA™ (hyaluronate sodium) Injectable Solution is a veterinary prescription treatment designed to support joint health in horses affected by osteoarthritis. Administered intravenously by a veterinarian, it helps relieve joint dysfunction in the carpus or fetlock associated with non-infectious synovitis. The sterile, preservative-free solution contains hyaluronate sodium, a naturally occurring joint lubricant that helps improve mobility and comfort.
NOTE: For generic medications, the product image shown may not reflect the actual item received. Generic medications can vary in size, color, and shape between orders depending on the manufacturer. Additionally, your medication may arrive packaged in a HardyPaw Pharmacy vial.
Benefits
- Joint Lubrication Support: Hyaluronic acid serves as a natural lubricant in synovial fluid. It helps keep joints in horses moving smoothly and flexibly.
- Anti-Inflammatory Action: Hyaluronate sodium can block the movement of inflammatory cells. This action helps reduce synovial inflammation related to osteoarthritis.
- Improves Joint Function: This treatment targets dysfunction in carpal and fetlock joints caused by non-infectious synovitis associated with equine osteoarthritis.
- Supports Mobility: Clinical studies showed good or excellent improvement in many horses after receiving intravenous hyaluronate therapy.
- Sterile Injectable Formula: Clear, pyrogen-free injectable solution produced through purified microbial extraction to ensure high purity and safety.
Variants
4 mL (40 mg) vial injectable solution
Ingredients
Composition Table
|
Ingredient |
Quantity |
|
Hyaluronate Sodium |
10 mg/mL |
|
Sodium Chloride |
8.5 mg/mL |
|
Sodium Phosphate Dibasic |
0.223 mg/mL |
|
Sodium Phosphate Monobasic |
0.04 mg/mL |
How to Use
Recommended-Dosage Chart
|
Horse Weight |
Dosage |
Frequency |
Route |
|
As directed by the veterinarian |
4 mL (40 mg) |
Once weekly for up to 3 treatments |
Intravenous injection |
Dosage & Administration Instructions
- Veterinary Prescription: NexHA™ is a prescription medication and must only be administered by or under the order of a licensed veterinarian.
- Recommended Dose: The manufacturer suggests giving 4 mL with 40 mg of hyaluronate sodium through an intravenous injection in horses.
- Injection Method: Inject the solution slowly into the jugular vein while using aseptic technique to keep it sterile.
- Treatment Schedule: You can repeat treatments weekly, up to a total of three injections.
- Post-Treatment Rest: Horses should remain on stall rest after treatment before gradually returning to normal activity levels.
Disclaimer: This dosage information is provided by the manufacturer. Always consult your veterinarian before administering or adjusting any supplement for your pet.
Additional Information
Precautions
- Radiographic Evaluation: Perform radiographic examination in cases of acute lameness to ensure joints are free from serious fractures.
- Breeding Safety: Safety has not been evaluated in breeding stallions or breeding, pregnant, or lactating mares.
- Human Consumption Warning: Do not administer to horses intended for human consumption as directed by the manufacturer.
- Route Restriction: This formulation is intended for intravenous use in horses and should not be administered intra-articularly.
Possible Side Effects
- Behavior Changes: Some reports indicate that horses may show occasional depression or lethargy after treatment during post-approval monitoring.
- Fever Episodes: A few horses might experience a short fever after getting hyaluronate sodium injectable treatment.
- Post-Injection Swelling: Mild temporary swelling can happen when hyaluronate is injected directly into joints during clinical studies.
Storage Information
- Temperature Control: Store NexHA injectable solution at temperatures below 25°C or 77°F to maintain product stability.
- Single-Use Vial: Discard any unused portion of the drug and the empty vial immediately after opening.
- Secure Storage: Keep this medication out of reach of children and store it with other veterinary drugs safely.
Shipping & Return
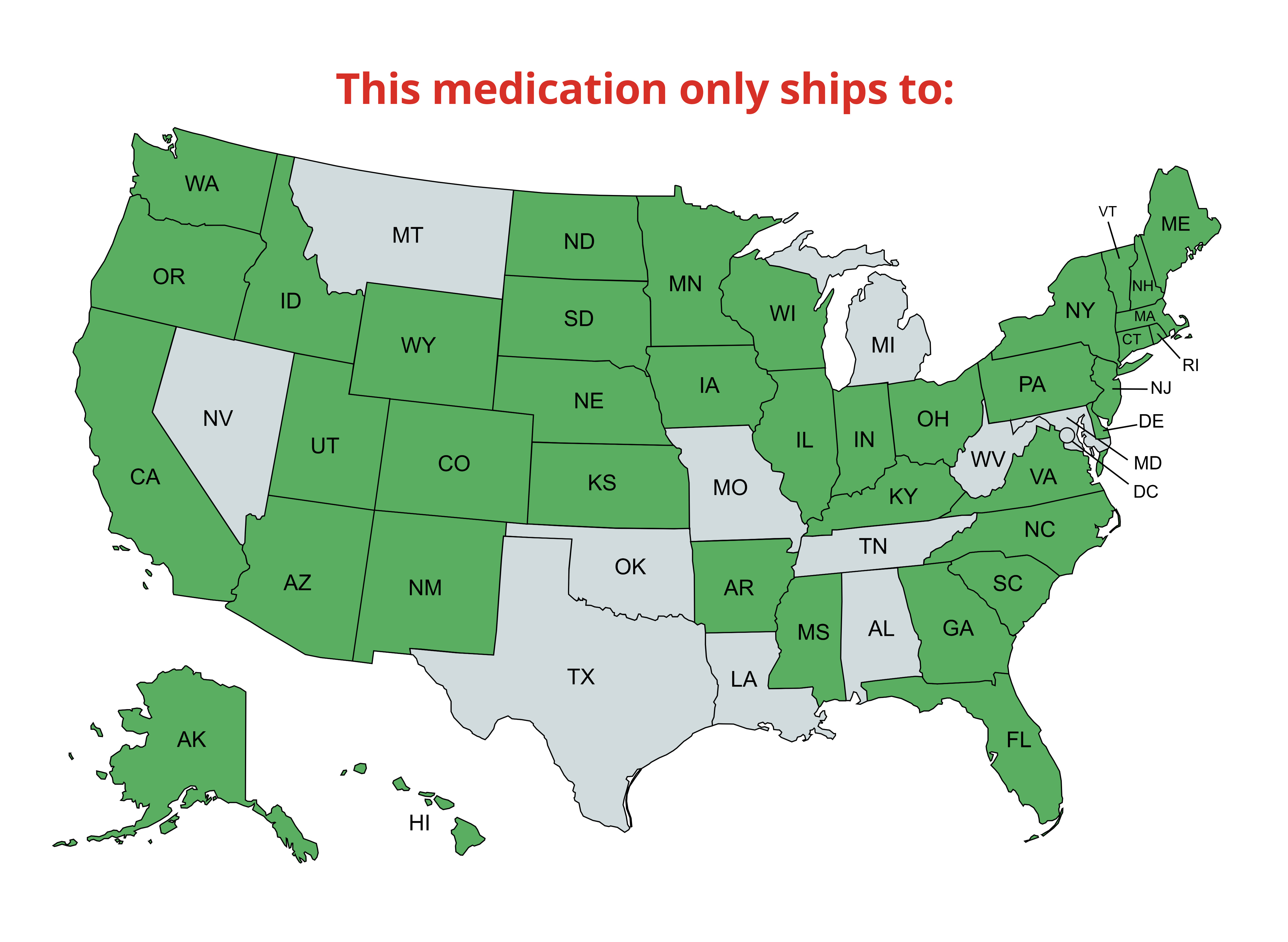
We offer ground, express, priority, and international delivery; see our shipping policy for details.
For return instructions or product concerns, please refer to our detailed refund policy.
Prescription items are NON-RETURNABLE and NON-REFUNDABLE.
Frequently Asked Questions
Q1: What is the NexHA™ injectable solution used for in horses?
Ans: NexHA™ injectable solution is used to treat joint dysfunction in horses, particularly in the carpal and fetlock joints. It is indicated for cases of non-infectious synovitis associated with equine osteoarthritis and helps support joint lubrication and mobility when administered intravenously by a veterinarian.
Q2: How does hyaluronate sodium help improve joint health in horses?
Ans: Hyaluronate sodium is a naturally occurring component of synovial fluid that helps maintain joint lubrication and viscoelasticity. It may also reduce inflammation by inhibiting certain inflammatory cells, which helps support joint function and comfort in horses affected by osteoarthritis.
Q3: How often can NexHA™ be administered to horses?
Ans: The recommended dose is 4 mL, which contains 40 mg of hyaluronate sodium given intravenously. You can repeat the treatments once a week for up to three total injections, based on how the horse responds and what the veterinarian advises. Always follow your veterinarian's instructions for the right treatment schedule.
Q4: Is NexHA™ safe for all horses?
Ans: NexHA™ has no known reasons not to use it in horses. However, its safety has not been checked in breeding stallions or in breeding, pregnant, or nursing mares. A veterinarian should evaluate the horse’s condition before treatment to make sure the product is suitable.
Q5: Are there any side effects associated with NexHA™ injections?
Ans: Clinical field studies reported no significant local or systemic side effects. However, voluntary post-approval reports have noted occasional depression, lethargy, and fever in some horses after treatment. If unusual symptoms appear, contact your veterinarian promptly for evaluation and guidance.



