Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Oral Lymphoma Treatment Tablets for Dogs
Product Description
LAVERDIA-CA1 (verdinexor tablets) is an orally administered medication for the treatment of lymphoma in dogs, and it must be used under veterinary guidance. Its mechanism involves selectively inhibiting nuclear export proteins that support the survival of cancer cells, thereby focusing on tumor cells that have genomic damage. It is given twice a week alongside food, offering a convenient at-home treatment option with specific tablet strengths tailored for individual dosing.
NOTE: For generic medications, the product image shown may not reflect the actual item received. Generic medications can vary in size, color, and shape between orders depending on the manufacturer. Additionally, your medication may arrive packaged in a HardyPaw Pharmacy vial.
Benefits
- Targets Lymphoma Cells: Selectively blocks the nuclear export protein CRM1, leading to the demise of cancer cells while largely protecting normal cells that possess healthy genomes.
- Oral Administration: Available in tablet form, which enables easy at-home dosing under the guidance of a veterinarian, eliminating the need for frequent visits to the clinic.
- Flexible Dosing: Various tablet strengths allow for adjustments in dosing according to the dog's weight, tolerance, and clinical response.
- Veterinary Guided Therapy: It is advisable to administer this medication under the supervision of a veterinarian, particularly those skilled in cancer treatment protocols for dogs.
- Food-Enhanced Absorption: Taking the medication with food enhances drug absorption and consistency, leading to more reliable therapeutic levels in the treated dogs.
Variants
50 mg Tablets
Ingredients
Verdinexor
How to Use
Recommended-Dosage Chart
|
Dog Weight (kg) |
Starting Dose (mg/kg) |
Frequency |
|
≥ 9 kg |
1.25 mg/kg |
Twice weekly with at least 72 hours between doses |
|
If tolerated after 2 weeks |
Increase to 1.5 mg/kg |
Twice weekly with at least 72 hours between doses |
|
Dose reduction if needed |
Minimum 1 mg/kg |
Twice weekly with at least 72 hours between doses |
Dosage & Administration Instructions
- Feed Before Dosing: Provide food immediately prior to administration because feeding improves absorption and helps achieve consistent therapeutic drug exposure.
- Wear Protective Gloves: Use disposable chemotherapy-resistant gloves while handling tablets to prevent accidental exposure to this antineoplastic medication.
- Avoid Crushing: Avoid dividing, crushing, or damaging tablets to maintain proper dosing and safe usage.
- Veterinary Use Only: This medication is available solely through a licensed veterinarian's prescription or direction.
- Observe Tolerance: Modifications in dosage or pauses in treatment may be necessary based on side effects and the veterinarian's evaluation during the course of treatment.
Disclaimer: This dosage information is provided by the manufacturer. Always consult your veterinarian before administering or adjusting any supplement for your pet.
Additional Information
Precautions
- Avoid Breeding Dogs: Due to potential fertility effects and teratogenic risks, do not use in pregnant, lactating, or breeding dogs.
- Health Monitoring Required: Frequent monitoring of bloodwork and serum chemistry is recommended during treatment due to potential abnormalities.
- Concurrent Conditions Caution: Safety has not been evaluated in dogs with serious infections, organ disease, diabetes, or other malignancies.
- Drug Interaction Awareness: Due to possible interaction, exercise caution when using it alongside medications that are metabolized through glutathione pathways, including acetaminophen.
Possible Side Effects
- Gastrointestinal Reactions: Typical reactions include vomiting, diarrhea, decreased appetite, and weight loss throughout the course of treatment.
- Lethargy: Dogs might show signs of lethargy, weakness, fever, or lowered activity levels while undergoing therapy.
- Blood Changes: Hematological alterations such as thrombocytopenia, anemia, or changes in leukocyte counts may develop and necessitate monitoring. Kidney or
- Liver Impact: Variations in kidney function indicators or liver enzyme levels have been noted during the treatment.
Storage Information
- Temperature: Store the medication between 20°C and 25°C to maintain the product's stability.
- Storage Safety: Medication must be kept in a secure place, away from children, pets, and other animals, to prevent accidental ingestion.
- Packaging: Retain the medication in its original bottle, which should have a secure cap and desiccant to protect against moisture.
Shipping & Return
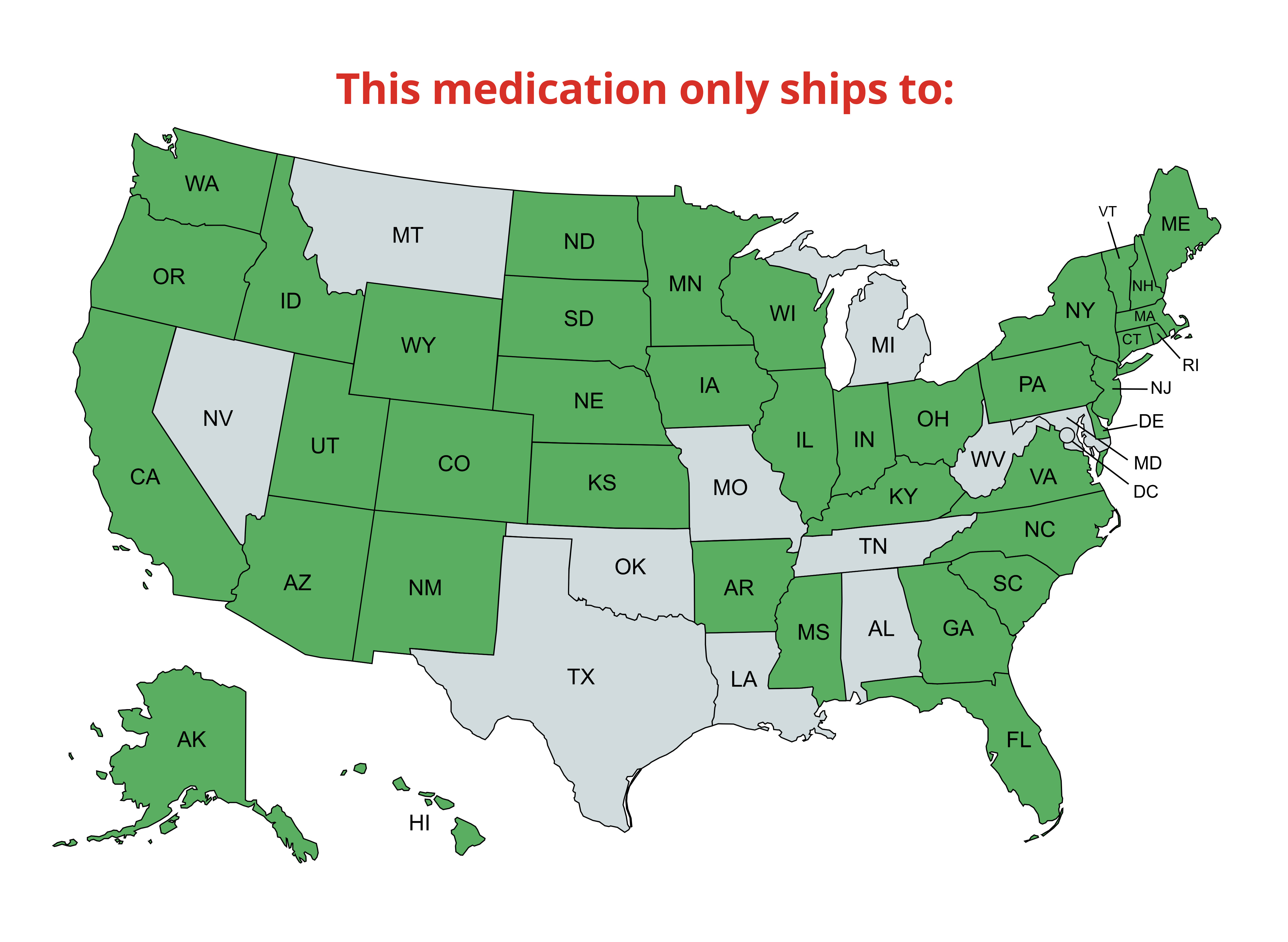
We offer ground, express, priority, and international delivery; see our shipping policy for details.
For return instructions or product concerns, please refer to our detailed refund policy.
Prescription items are NON-RETURNABLE and NON-REFUNDABLE.
Frequently Asked Questions
Q1: What is LAVERDIA-CA1 used for in dogs?
Ans: LAVERDIA-CA1 is prescribed for the treatment of lymphoma in dogs under veterinary supervision. It contains verdinexor, an inhibitor of nuclear export proteins, which contributes to the growth of cancer cells. It specifically targets tumor cells with genetic damage while preserving healthy cells.
Q2: How is LAVERDIA-CA1 administered to dogs?
Ans: LAVERDIA-CA1 is given orally twice a week, with at least 72 hours between each dose. Feed dogs immediately prior to administration to enhance the absorption of the medication. The tablets must not be split or crushed, and the dosage should consistently follow the veterinarian's guidance.
Q3: Is LAVERDIA-CA1 a chemotherapy drug?
Ans: LAVERDIA-CA1 is an antineoplastic drug utilized in the field of veterinary oncology. It is categorized as a selective inhibitor of nuclear export, specifically targeting cancerous cells as it functions as a chemotherapy agent. Always use protective handling measures, such as wearing gloves, during its administration.
Q4: What side effects can occur with LAVERDIA-CA1 treatment?
Ans: Typical side effects are vomiting, diarrhea, reduced appetite, weight loss, and fatigue. Additionally, there might be changes in blood counts and fluctuations in kidney or liver function tests. Regular veterinarian assessment is advised to effectively address any negative reactions.
Q5: Who should not handle LAVERDIA-CA1 tablets?
Ans: Expectant mothers, women who might become pregnant, and those who are breastfeeding should avoid handling LAVERDIA-CA1 or touching dog waste that has been treated due to possible risks to reproduction. Anyone giving the medication must wear gloves that are resistant to chemotherapy to prevent exposure.



