Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Oral Canine Lymphoma Treatment Tablets
Product Description
LAVERDIA-CA1 is a prescription cancer medication for dogs that supports the treatment of lymphoma through a targeted mechanism involving verdinexor, its active ingredient. This orally administered coated tablet works by blocking transport proteins so tumor-suppressing proteins remain inside cancer cells, disrupting cell survival and helping to reduce disease progression in canine lymphoma under veterinary care.
NOTE: For generic medications, the product image shown may not reflect the actual item received. Generic medications can vary in size, color, and shape between orders depending on the manufacturer. Additionally, your medication may arrive packaged in a HardyPaw Pharmacy vial.
Benefits
- Targeted Cancer Support: Uses verdinexor to help disrupt lymphoma cell survival pathways in dogs with diagnosed lymphoma.
- Oral Administration: Easy to give tablets by mouth according to the prescribed dosing schedule.
- Multiple Strengths: Available in 2.5 mg, 10 mg, and 50 mg tablet strengths for precise dosing.
- Conditional FDA Approval: Approved for safe use in canine lymphoma treatment based on a reasonable expectation of effectiveness.
Ingredients
Active Ingredients
Verdinexor
How to Use
Dosage & Administration Instructions
- Prescription Requirement: LAVERDIA-CA1 is labeled for use only as directed by a licensed veterinarian and under veterinary oversight.
- Fed Conditions: It is approved for use with a small meal immediately before administration to support absorption.
- Beginning Dosage Amount: The product label suggests starting at 1.25 mg for every kilogram of body weight, given twice a week, with no less than 72 hours between each dose.
- Dose Adjustment Scope: Labeled dosing may be increased to 1.5 mg/kg twice weekly after two weeks if well tolerated.
- Reduction Guidelines: Labeled dose reductions to 1 mg/kg twice weekly are permitted based on adverse reactions.
Disclaimer: This dosage information is provided by the manufacturer. Always consult your veterinarian before administering or adjusting any medication for your pet.
Additional Information
Precautions
- Pregnancy Risk: Use is contraindicated in pregnant, lactating, or breeding dogs due to potential reproductive effects.
- Concurrent Diseases: Safe use in dogs with serious infections or major organ disease has not been evaluated.
- Drug Interactions: The effect of other medications on the metabolism of this product is not fully established.
- Young Dogs: Safety in dogs younger than seven months of age has not been evaluated.
Possible Side Effects
- Decreased Appetite: Loss of appetite (anorexia) was commonly observed in dogs treated with verdinexor.
- Gastrointestinal Reactions: Vomiting and diarrhea were among the frequently reported reactions in treated dogs.
- Weight Loss: Weight reduction was noted as an adverse effect during studies.
- Lethargy Reports: Lethargy and weakness appeared among recorded adverse reactions.
Storage Information
- Room Temperature Storage: The product should be kept at normal room temperature, ideally between 20 °C and 25 °C, which equals 68 °F to 77 °F.
- Secure Location: It is recommended to keep the medication out of reach of children and animals to prevent accidental access.
- Sealed Containers: The product is supplied in child-resistant, heat-sealed bottles with desiccant to maintain stability.
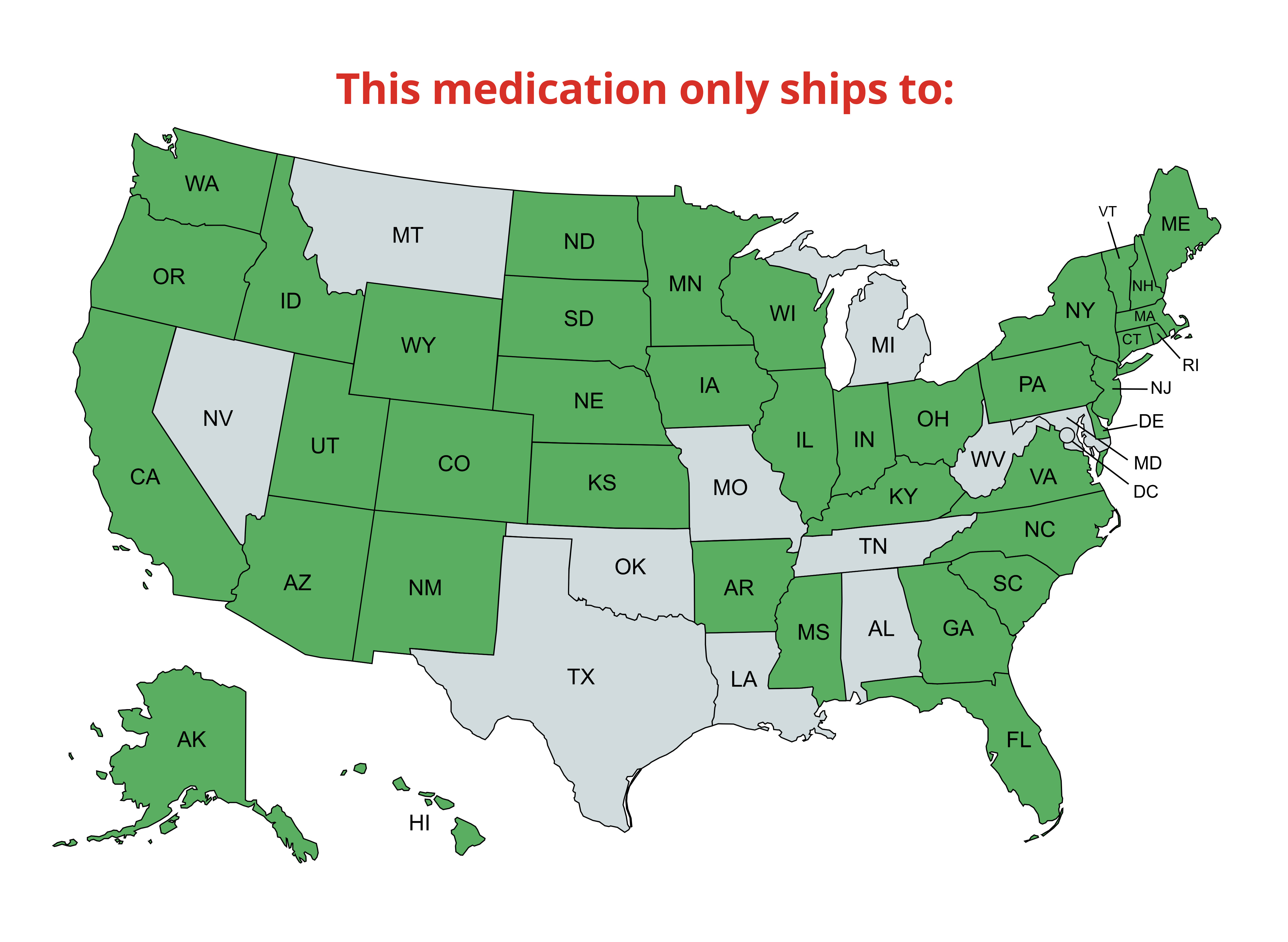
Shipping & Return
We offer ground, express, priority, and international delivery; see our shipping policy for details.
For return instructions or product concerns, please refer to our detailed refund policy.
Prescription items are NON-RETURNABLE and NON-REFUNDABLE.
Frequently Asked Questions
Q1: What is LAVERDIA-CA1 used for in dogs?
Ans: LAVERDIA-CA1 is a prescription medication that is conditionally FDA-approved for the treatment of lymphoma in dogs. It contains verdinexor, which targets cancer cell survival mechanisms and is intended for use only under licensed veterinary supervision.
Q2: How does LAVERDIA-CA1 work in canine lymphoma?
Ans: LAVERDIA-CA1 works by inhibiting a protein responsible for exporting tumor suppressor proteins from the cell nucleus. By retaining confirms within the nucleus, verdinexor disrupts cancer cell survival pathways and selectively affects cells with genomic damage.
Q3: Is LAVERDIA-CA1 approved by the FDA?
Ans: LAVERDIA-CA1 is conditionally approved by the FDA for the treatment of lymphoma in dogs. Conditional approval indicates a reasonable expectation of effectiveness while additional clinical studies are conducted to fully demonstrate therapeutic benefit.
Q4: What are the most common side effects of LAVERDIA-CA1?
Ans: The most commonly reported side effects in dogs treated with LAVERDIA-CA1 include decreased appetite, vomiting, diarrhea, weight loss, and lethargy. More serious reactions have also been reported, making regular veterinary monitoring an essential part of treatment.
Q5: Does LAVERDIA-CA1 require special handling precautions?
Ans: Yes, LAVERDIA-CA1 is an antineoplastic drug and requires special handling precautions. Manufacturer labeling recommends protective gloves during handling and limits exposure for pregnant or nursing individuals due to potential reproductive and safety risks.