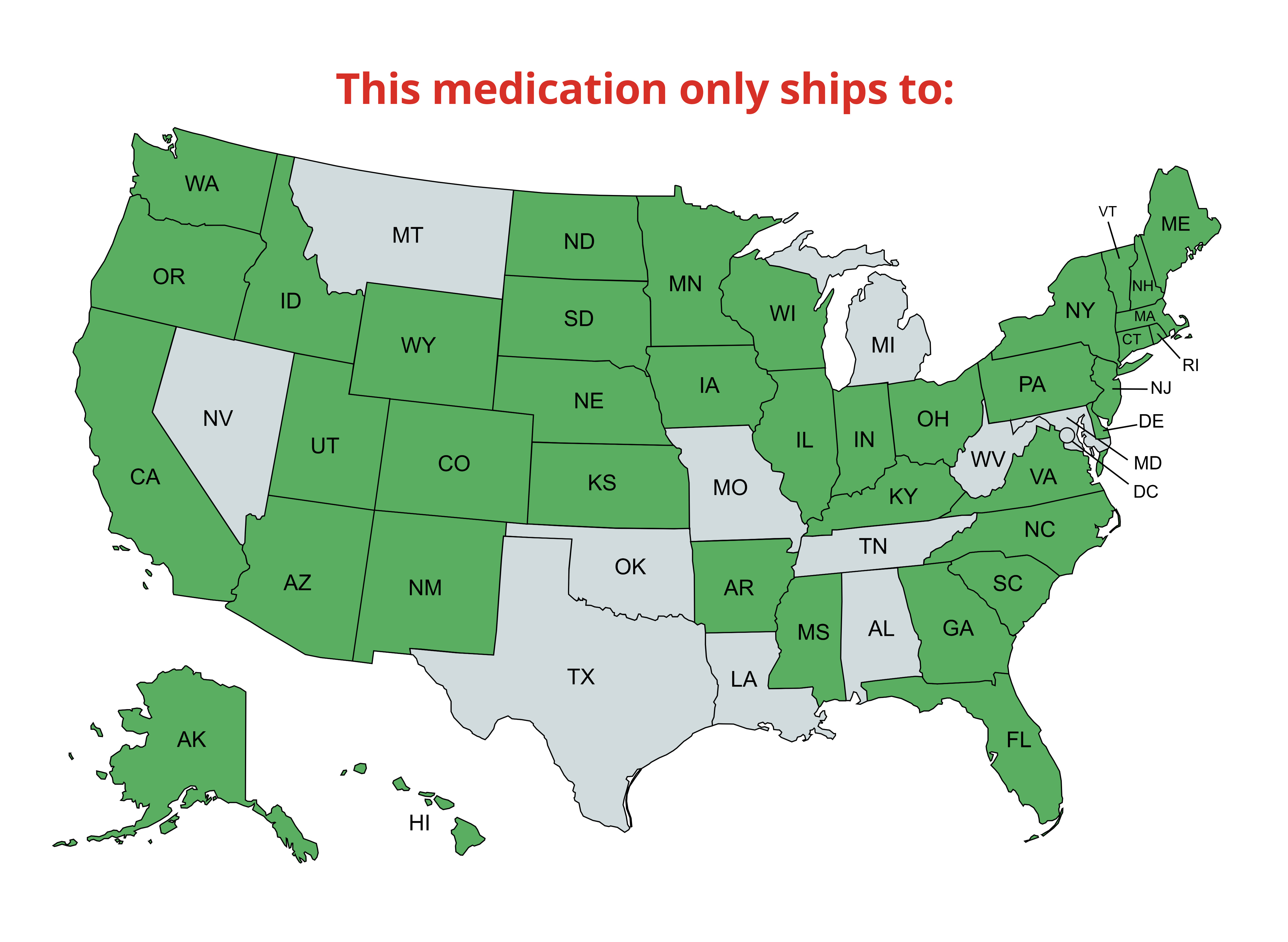
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Oral Lymphoma Treatment Tablets for Dogs Support
Product Description
Laverdia-CA1 is an orally administered cancer medication indicated for lymphoma treatment in dogs. It contains verdinexor, which inhibits the growth of cancer cells by interfering with nuclear export proteins. This treatment has received conditional authorization from the U.S. Food and Drug Administration for veterinary use under the supervision of a qualified professional.
NOTE: For generic medications, the product image shown may not reflect the actual item received. Generic medications can vary in size, color, and shape between orders depending on the manufacturer. Additionally, your medication may arrive packaged in a HardyPaw Pharmacy vial.
Benefits
- Targets Cancer Cells: Selectively inhibits nuclear export protein CRM1, helping retain tumor suppressor proteins in the nucleus to limit cancer progression.
- Oral Administration: The tablet formulation enables dosing at home under veterinary supervision, reducing the need for frequent visits to the clinic for injectable chemotherapy treatments.
- Flexible Dosing Schedule: Typically given twice a week, with dose modifications based on the patient's tolerance, allowing veterinarians to customize therapy to meet individual patient requirements.
- Clinically Evaluated: Field studies confirming conditional FDA approval have shown a measurable response in dogs with newly diagnosed or relapsed lymphoma.
-
Convenient Strength Options: Offered in various tablet strengths to facilitate precise weight-based dosing for a broad spectrum of dog sizes.
Ingredients
Verdinexor 2.5 mg per tablet
How to Use
Recommended-Dosage Chart
|
Dog Weight (kg) |
Initial Dose (mg/kg) |
Frequency |
Notes |
|
≥ 9 kg |
1.25 mg/kg |
Twice weekly |
Minimum 72 hours between doses. |
|
After 2 weeks, if tolerated |
1.5 mg/kg |
Twice weekly |
Dose increases based on tolerance. |
|
If adverse reactions occur |
1 mg/kg minimum |
Twice weekly |
Dose reduction or interruption may be required. |
Dosage & Administration Instructions
- Feed Before Dosing: Administer tablets immediately after feeding to improve drug absorption and achieve more consistent therapeutic exposure levels.
- Twice Weekly Schedule: Give orally two times per week with at least seventy-two hours between doses as directed.
- Do Not Crush: Tablets should not be split, crushed, or broken to maintain accurate dosing and safety during administration.
- Protective Handling: Wear chemotherapy-resistant disposable gloves when handling tablets to avoid accidental exposure to the medication.
- Veterinary Prescription: This medication is restricted to use by or on the order of a licensed veterinarian only.
Disclaimer: This dosage information is provided by the manufacturer. Always consult your veterinarian before administering or adjusting any supplement for your pet.
Additional Information
Precautions
- Health Monitoring: Dogs should be monitored frequently for blood and serum chemistry abnormalities during treatment initiation and maintenance periods.
- Medical Conditions: Safety has not been evaluated in dogs with serious infections, organ disease, diabetes, or clinically relevant hypercalcemia.
- Breeding Restrictions: Do not use in pregnant, lactating, or breeding dogs due to potential fertility effects and developmental risks.
- Drug Interactions: Concurrent use with medications undergoing glutathione conjugation metabolism should be minimized to avoid metabolic interference risks.
- Young Dogs: Safety has not been established in dogs younger than seven months of age, according to manufacturer information.
Possible Side Effects
- Gastrointestinal Signs: Reduced appetite, nausea, diarrhea, and loss of weight are some of the side effects that might require supportive care from a veterinarian.
- Lethargy: During treatment, dogs might experience fatigue, weakness, or elevated temperature, as observed in clinical studies.
- Blood Abnormalities: Changes such as thrombocytopenia, anemia, or leukocyte abnormalities have been reported during treatment in some dogs.
- Kidney Changes: Increased thirst, urination abnormalities, or urinary infections may occur as noted in treated dogs.
- Liver Effects: Elevated liver enzymes or bilirubin-related changes have been observed in some treated animals.
Storage Information
- Room Temperature: Maintain a stable temperature of 20°C to 25°C for best stability.
- Original Container: Store in the original container with a desiccant and a child-proof cap to ensure product quality.
- Secure Location: Keep it in a safe place away from pets, children, food prep areas, and other human medications.
Shipping & Return
We offer ground, express, priority, and international delivery; see our shipping policy for details.
For return instructions or product concerns, please refer to our detailed refund policy.
Prescription items are NON-RETURNABLE and NON-REFUNDABLE.
Frequently Asked Questions
Q1: What is Laverdia-CA1 used for in dogs?
Ans: Laverdia-CA1 is an oral anticancer drug prescribed for dogs with lymphoma. It contains verdinexor, which inhibits proteins that allow cancer cells to grow. The FDA has conditionally approved this therapy for veterinary use under the guidance of a professional.
Q2: How is Laverdia-CA1 administered to dogs?
Ans: The medication is given orally, typically twice per week with at least seventy-two hours between doses. Dogs should be fed before administration to improve absorption. Tablets must not be crushed or split, and dosing adjustments are made by a veterinarian based on tolerance.
Q3: How effective is Laverdia-CA1 for canine lymphoma?
Ans: Clinical studies in the field showed an objective response rate of about thirty-four percent in dogs with newly diagnosed or relapsed lymphoma. The median time to disease progression varied, with some dogs having longer responses based on individual factors and how well they tolerated treatment.
Q4: What side effects should pet owners watch for?
Ans: Frequent side effects can include reduced appetite, nausea, diarrhea, fatigue, and weight loss. There have also been changes in blood parameters, kidney function, and raised liver enzymes. Canines in treatment should be monitored closely by a veterinarian for any troubling clinical signs.
Q5: Are there any safety precautions for handling this medication?
Ans: Yes, protective chemotherapy-resistant gloves should be worn when handling tablets or waste from treated dogs. Pregnant or nursing individuals should avoid contact and should store away from the reach of children to avoid accidental exposure.


