Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
Product title
Vendor
$19.99 | $24.99
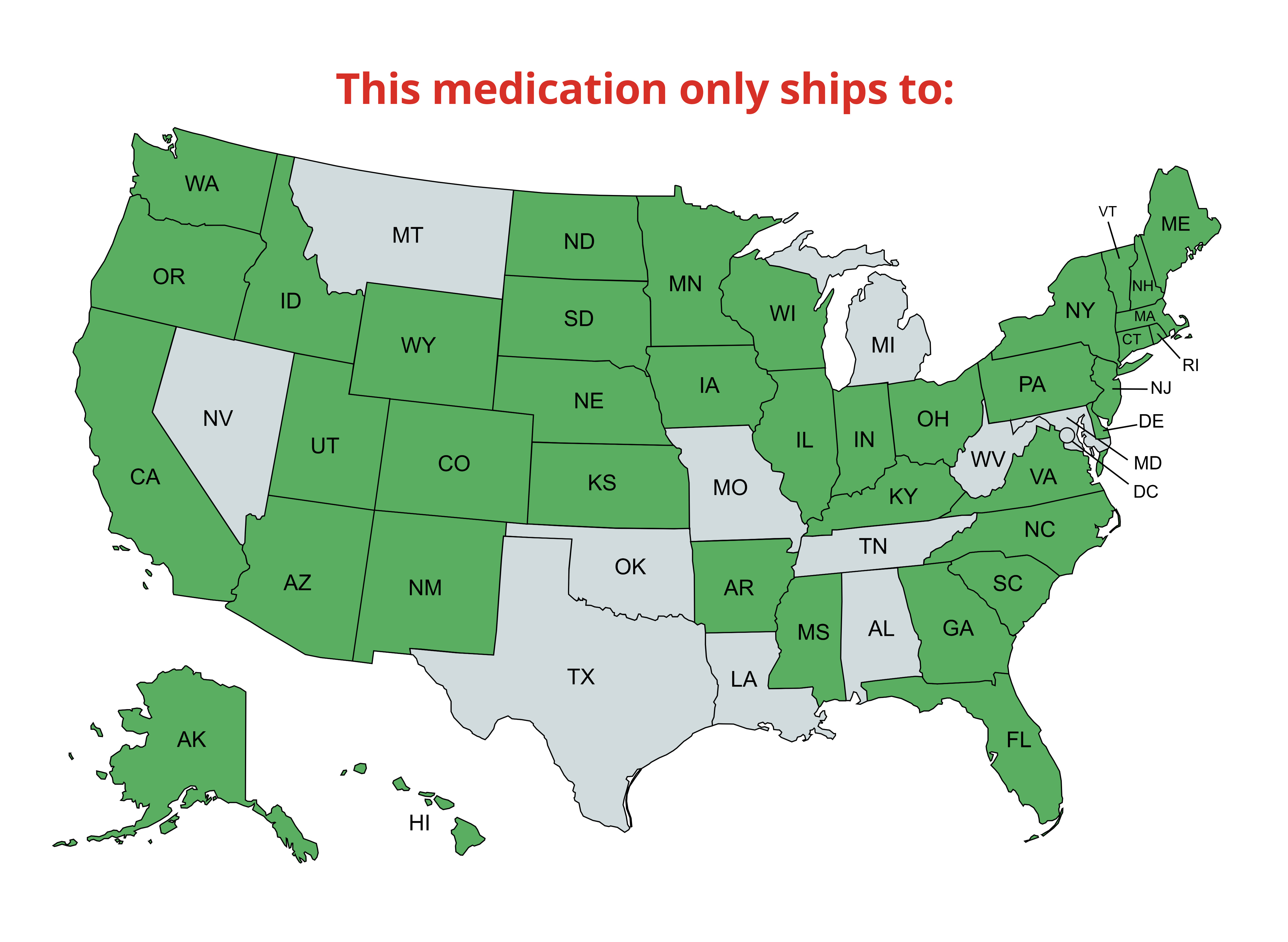
Treatment for Bovine Respiratory Disease in Cattle
Product Description
Draxxin® KP is a prescription injectable treatment for cattle that addresses bovine respiratory disease caused by Histophilus somni, Pasteurella multocida, Mannheimia haemolytica, and Mycoplasma bovis, in addition to managing fever associated with BRD. It combines tulathromycin, a macrolide antimicrobial, with ketoprofen, a nonsteroidal anti-inflammatory drug, in a single subcutaneous dose for effective treatment and rapid pyrexia reduction.
Benefits
- Dual Action Formula: Combines tulathromycin and ketoprofen to treat bacterial BRD and control associated fever in cattle.
- Single Dose Therapy: Administered once subcutaneously at the labeled dosage for convenient BRD treatment and pyrexia control.
- Proven BRD Effectiveness: Demonstrated significantly higher treatment success compared to saline in a multi-site field study.
- Rapid Fever Reduction: Showed significant temperature reduction within six hours compared to tulathromycin alone.
- FDA Approved Use: Authorized by the FDA as both safe and effective when used following the instructions on the label.
Variants
50 mL
100 mL
250 mL
500 mL
Ingredients
Composition Table
|
Ingredient |
Quantity |
|
Tulathromycin |
100 mg/mL |
|
Ketoprofen |
120 mg/mL |
How to Use
Recommended-Dosage Chart
|
Animal Species |
Dosage |
Route |
Frequency |
|
Cattle |
2.5 mg/lb tulathromycin and 3 mg/lb ketoprofen, equivalent to 1.1 mL per 100 lb body weight |
Subcutaneous injection |
Single dose |
Dosage & Administration Instructions
- Single Subcutaneous Dose: Administer once subcutaneously at 1.1 mL per 100 lb body weight.
- Injection Site Limit: Do not inject more than 10 mL per injection site in cattle.
- Body Weight Accuracy: Determine accurate body weight before treatment to ensure proper dosage calculation.
- Prescription Required: This medication is limited by federal regulation to be utilized only by, or under the directive of, a licensed veterinarian.
Disclaimer: This dosage information is provided by the manufacturer. Always consult your veterinarian before administering or adjusting any supplement for your pet.
Additional Information
Precautions
- Not Intended for Dairy Cattle: Usage is limited to female dairy cattle that are 20 months old or older.
- Calves For Veal: Do not use in calves intended for veal production.
- Withdrawal from Slaughter: Cattle are prohibited from being slaughtered for consumption for 18 days following treatment.
- Human Safety Warning: Not for human use. Keep out of reach of children.
Possible Side Effects
- Injection Site Reactions: Treated cattle may experience temporary swelling and inflammation at the injection site.
- Gastrointestinal Effects: The NSAID component could lead to gastrointestinal irritation in animals that are sensitive.
- Hypersensitivity Risk: Animals that are sensitive to macrolides or NSAIDs may have allergic reactions.
Storage Information
- Controlled Temperature: Store at 20°C to 25°C with excursions permitted between 15°C and 30°C.
- Protect from Light: Keep the bottle in its original packaging to protect from light exposure.
- Use Period: Use within 56 days of first vial puncture.
Shipping & Return
We offer ground, express, priority, and international delivery; see our shipping policy for details.
For return instructions or product concerns, please refer to our detailed refund policy.
Prescription items are NON-RETURNABLE and NON-REFUNDABLE.
Frequently Asked Questions
Q1: What is Draxxin KP used for in cattle?
Ans: Draxxin KP is indicated for the treatment of bovine respiratory disease related to Mannheimia haemolytica, Pasteurella multocida, Histophilus somni, and Mycoplasma bovis. It is additionally authorized for managing fever related to bovine respiratory illness in cattle.
Q2: How is Draxxin KP administered to cattle?
Ans: Draxxin KP is administered as a single subcutaneous injection at a dosage of 1.1 mL per 100 lb body weight. No more than 10 mL should be injected per site. Accurate body weight determination is essential for proper dosing.
Q3: What is the slaughter withdrawal period for Draxxin KP?
Ans: Cattle treated with Draxxin KP must not be slaughtered for food within 18 days following treatment. This withdrawal period is established to ensure drug residues remain within approved tolerance levels for food safety compliance.
Q4: Can Draxxin KP be used in dairy cattle or veal calves?
Ans: Draxxin KP is not recommended for use in female dairy cattle that are 20 months old or older. It is also not approved for use in calves intended for veal production, according to the FDA-approved labeling.
Q5: Is Draxxin KP available without a prescription?
Ans: Draxxin KP is a prescription veterinary medication. Federal law restricts its use to licensed veterinarians or by their order. It should only be administered under professional veterinary supervision to ensure safe and appropriate use.





